We present a comprehensive overview of the trials of apollo book three the burning maze. This comprehensive guide covers the essential aspects and latest developments within the field.
the trials of apollo book three the burning maze remains a foundational element in understanding the broader context. Our automated engine has curated the most relevant insights to provide you with a high-level overview.
"the trials of apollo book three the burning maze represents a significant milestone in our collective understanding of this niche."
Below you will find a curated collection of visual insights and related media gathered for the trials of apollo book three the burning maze.
Curated Insights
Click on trial number to go to the associated ClinicalTrials.gov webpage, to view the protocol title and study information. This portfolio shows NCTN trials. For information about NCORP trials, please see: …
Assay sensitivity is the ability of the trial to have detected a difference between treatments of a specified size. Historically, have consistent findings been observed with the active control...
Randomized clinical trials are scientific investigations that examine and evaluate the safety and efficacy of new drugs, devices, tests, or lifestyle interventions using human subjects.
These clinical trials — from tuberculosis and Lassa fever to cancer and autoimmune disorders — offer a glimpse into a future in which precision medicine, innovation and persistence could ...
Increasing patient engagement in clinical trials offers opportunities to improve enrollment and retention in clinical trials, helping ensure interventions can be evaluated effectively and that clinical research …
Apr 28, 2026 · • The pilot will involve the recruitment of sponsors that are currently or will be pursuing early phase clinical trials through product applications submitted to the Center for Drug Evaluation …
Jun 27, 2018 · The final rule for Clinical Trials Registration and Results Information Submission (42 CFR Part 11) specifies requirements for submitting clinical trial information to ClinicalTrials.gov.
Captured Moments

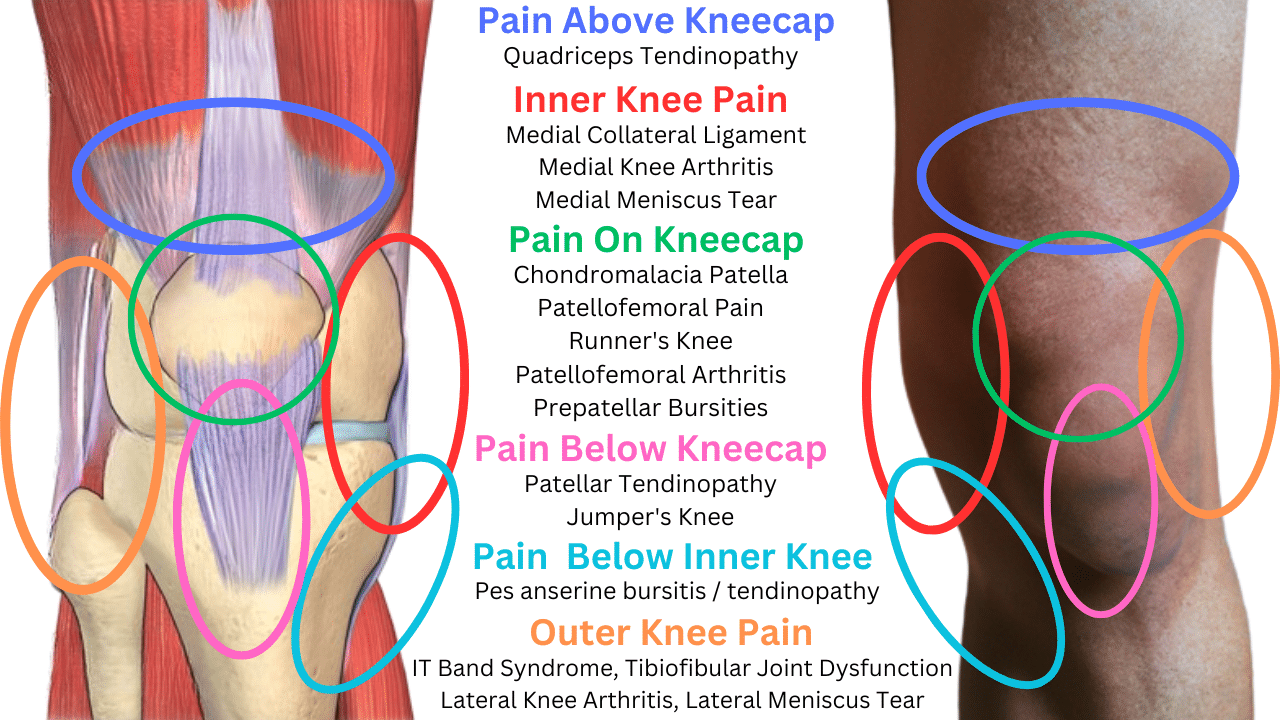
Lower Lateral Knee Pain